When people initially start digging for their own root cause of Hashimoto’s, they will usually find more than one, and some of those may prove to be more elusive than others. Some that are commonly found include food sensitivities (such as gluten and dairy), chronic infections, intestinal permeability, environmental toxins, and even ongoing, everyday stress.
One root cause that is always on the top of my list when I begin working with a new client, is related to nutrient deficiencies. Low ferritin anemia seems to be a pervasive issue in those with Hashimoto’s. When I first learned about iron deficiency in pharmacy school, the condition didn’t seem like rocket-science. You have low iron? Get more iron in your diet and/or take an iron supplement! Easy. But if things were only this simple… 🙂
Iron deficiency anemia (also known simply as iron deficiency) caused one of the first symptoms that I noticed in my own Hashimoto’s journey: hair loss! My lion’s mane was coming out while shampooing, brushing, walking, and talking – and as a Leo, you can bet I was alarmed. I was tested for anemia, but not “anemic” (per the usual tests), and eventually I learned that beyond the usual tests for anemia, there’s a more sensitive test for iron deficiency anemia known as ferritin, that is very relevant in Hashimoto’s and hair loss.
In fact, most people who are diagnosed with Hashimoto’s will present with low levels of ferritin, the iron storage protein that is important for proper thyroid function – but of course, most don’t get tested properly.
I commonly see iron deficiency – with its symptoms of fatigue, restless legs, insomnia, cold intolerance, moodiness, and the upsetting “thyroid” hair loss results – in many of my clients, even after they start thyroid medications like levothyroxine, Synthroid, and even Tirosint.
Not only is iron deficiency often missed because one of the most important tests for iron deficiency isn’t a routine test, but it can be more challenging to correct than simply taking a supplement. While dietary adjustments and supplementation can definitely help, time and again, I see it’s not so easy for some people (and yes, it’s true – I am “some people”, too)!
Sometimes we need to dig much deeper to truly uncover the root causes of iron deficiency, as well as try some more aggressive interventions.
In full disclosure, I sort of dove into a rabbit hole with this topic. I started doing research for myself and my clients who, despite eating steaks and burgers and taking iron supplements, didn’t see their iron levels budge – and lo and behold, the rabbit hole just kept getting deeper and deeper.
I have been writing this article for almost 10 years, and keep finding new and more reasons and solutions for iron issues, so I think you will definitely discover something new. I promise to list out the most common solutions first, but also share some insights if you’ve been struggling to get your ferritin levels up by trying all of the usual things. 🙂
I call it the “digging for the root cause, while chopping down branches” approach. I wish I had had this information when I was losing so much hair, feeling cold, moody, and tired!
Raising iron levels can be tricky for a number of reasons…
Some people can’t absorb iron despite an adequate diet.
Some people don’t tolerate iron supplements.
Some people may have adverse reactions to iron.
Some people don’t absorb or utilize iron because of other deficiencies.
Some people have heavy periods, hormonal imbalances, and even gut issues that rapidly deplete iron levels.
Even if you have all of these, I’m going to share with you how to address your iron deficiency.
In this article, you will discover:
- Anemias commonly found in Hashimoto’s
- Symptoms associated with iron deficiency anemia
- The causes of iron deficiency anemia in Hashimoto’s
- Recommended testing for ferritin (stored iron) levels
- The root cause approach to improving iron deficiency
Anemias Commonly Found in Hashimoto’s
You may have heard the term “anemia” kicked around a time or two, and most people associate it with iron deficiency, but that is technically not correct, as various issues can cause anemia. Anemia by definition is having a low level of red blood cells. The body needs red blood cells in order to get oxygen from the lungs, to the rest of the body. Red blood cells carry hemoglobin, a complex protein that contains iron molecules. It is these molecules that transport oxygen throughout the body.
I have found that anemia is very common in people with Hashimoto’s, and research has also found anemia to frequently occur with thyroid disease. [1]
There are actually three main types of nutrient-specific anemias commonly found in people with Hashimoto’s – low levels of vitamin B12, folate, or iron can all lead to anemia. (Note: a person can be deficient in any of these nutrients and not have anemia, but as a deficiency continues, anemia may develop.)
Today, we’ll be focusing on iron deficiency anemia (for more detailed information on the other two types of anemias, please see my B12 and MTHFR articles).
Iron deficiency anemia is very common in Hashimoto’s, and one recent study found that out of 144 hypothyroid patients, 102 were found to be deficient in iron. [2] That’s over 70 percent!
Iron Deficiency and Ferritin
Some people I’ve talked to mention that they’ve been tested, and do not have anemia, only to find out that they didn’t get the right tests.
Your doctor may test for anemia (low iron levels) by running a panel for red blood cells, hemoglobin, hematocrit, and iron levels, and all of them may come up normal. However, you may still be iron deficient if your iron stores are low (ferritin is the iron storage protein that can be tested). If not enough iron is available, the body may pull the iron from less important physiological processes, such as hair growth, to keep enough iron circulating in the blood.
Ferritin, the name given to the body’s iron reserve protein, is required for the transport of T3 to cell nuclei and the utilization of the T3 hormone. A decrease in ferritin can present as increased hair loss during shampooing and brushing, as well as overall thinning of hair without a specific pattern or bald spots.
Ferritin levels can also be measured and will give us an idea of how much iron is stored in the body and that is available for use. Ferritin should be checked in all women with Hashimoto’s and for anyone experiencing hair loss (as well as women who are pregnant, breastfeeding, menstruating, endurance athletes, or vegetarians; and men and postmenopausal women – so everyone, haha).
Normal ferritin levels for women are between 20 and 200 ng/mL. According to some experts, ferritin levels of at least 40 ng/mL are required to stop hair loss, while levels of at least 70 ng/mL are needed for hair regrowth. The optimal ferritin level for thyroid function is between 90-110 ng/mL.
You can check your ferritin levels easily with Ulta Lab Tests.
Iron deficiency anemia (also referred to as having a deficiency in ferritin, as the body’s iron reserve is stored as ferritin) is one of the most common nutrient deficiencies I see in people with Hashimoto’s.
As an essential mineral, iron is important for many reasons related to thyroid metabolism. Iron transports and affects the body’s utilization of T3. When ferritin levels are low, this can cause elevated reverse T3 levels, which blocks receptors for free T3 (our active thyroid hormone), resulting in thyroid symptoms. [3] Low iron may also reduce the binding ability of free T3. [4] This can occur even if someone is on levothyroxine, leading to that common dilemma of having thyroid symptoms, despite taking thyroid medications.
Additionally, some studies suggest that iron deficiency can inhibit the conversion of T4 to the active T3 thyroid hormone. [5]
A 2018 study found that in women with persistent symptoms of hypothyroidism despite levothyroxine therapy, two-thirds were able to eliminate their symptoms when they got their ferritin levels over 100 µg/l. [6]
Personally, I battled with low ferritin levels for a long time. My levels were jumping around between 8-16 µg/l. I would work really hard to get them up to 16 µg/l, then I would get my period and they would be 12 µg/l. I could not get them in the optimal zone despite eating liver, taking liver pills, taking cod liver oil, and eating red meat three times per day! One workaround for me to feel good despite low ferritin levels, was to take T3 hormone directly to “bypass” the low T3 levels and high reverse T3 levels caused by low ferritin (a.k.a. ongoing thyroid symptoms like hair loss and fatigue). I know many thyroid patients that have discovered they feel better on T3-containing meds, have never had their ferritin tested and don’t realize that ferritin is such an important reason that their T4-containing meds may not work properly.
There have been many studies correlating abnormal thyroid hormone levels and iron deficiency. For example, a 2017 study found that the thyroid hormone levels of anemic women with impaired thyroid function improved when they were given iron supplementation. [7]
Multiple studies have examined patients with coexisting iron-deficiency anemia and subclinical hypothyroidism, and found that the combination of iron and levothyroxine produced a larger increase in serum iron and blood count variables, than control groups who only received monotherapy of either iron or levothyroxine alone. [8]
A more recent 2019 study looked at the levels of three important nutrients for thyroid health: iodine, iron, and selenium. [9] Researchers found that Hashimoto’s patients were frequently iron-deficient due to co-morbidities of a condition called autoimmune gastritis, as well as celiac disease; and that many people with Hashimoto’s were also deficient in selenium. In their study, two-thirds of women with hypothyroidism – even while on levothyroxine medication – had symptom relief when their ferritin levels were restored above 100 µg/l (they also found that selenium can reduce TPO-antibody concentration, postpartum thyroiditis, and hypothyroidism).
On top of being common in those with Hashimoto’s, iron deficiency is very prevalent in women of childbearing age (20-50 percent of women, depending on the studies), those who are pregnant (some 50-80 percent of pregnant women), and women who are recently postpartum. [10] Breastfeeding, too, can deplete iron stores. [11] One study found that iron deficiency during early pregnancy could even lead to hypothyroidism. [12]
The good news is that, in most cases, improving iron levels has been shown to improve thyroid hormone levels as well as reduce many symptoms. We’ll talk more about how to specifically address iron deficiency, but first let’s take a moment to look at the symptoms of this condition.
Symptoms Associated with Iron Deficiency Anemia
Signs and symptoms of iron deficiency anemia may include: [13]
- Hair loss
- Insomnia
- Breathlessness
- Pale skin
- Easy bruising
- Dark circles under eyes
- Fatigue
- Upward curvature of the nails (koilonychia)
- Cracks in the corner of one’s mouth
- Hunger cravings for strange substances (ice, carrots… even paper!)
- Pale coloring, rather than pink, if you pull down your eyelid
- Restless legs and/or leg cramps
- Dizziness
- Crying, moodiness, and anxiety (that’s what happened to me during my pregnancy!)
I personally believe that some cases of depression are actually iron deficiency, especially in those who follow a vegan/vegetarian diet. Emily Deans, a psychiatrist who studies the link between food and mood, has observed this in her practice as well. [14] She has worked with numerous vegetarians who think they are having panic attacks, when it turns out they’re deficient in iron.
We can see that deficiencies in iron can make us feel less than optimal.
So you may ask, “Why not just eat more red meat?” or “Why don’t we all just go out and take a bunch of iron supplements?”
Unfortunately, in the case of iron, too much of a good thing is not good for you!
You really need to initially test your ferritin levels, and then monitor them over time. Additionally, it is often not enough to just eat more iron-rich foods or supplement with iron; many people may also need to address “why” they have iron deficiency in the first place.
Comprehensive Testing for Anemia
If your doctor suspects you may have iron deficiency anemia, he or she will likely run a panel for red blood cells to check your hemoglobin, hematocrit, and iron levels. This test, however, is really better suited for verifying advanced cases of anemia rather than for identifying earlier stages of thyroid relevant nutrient depletions.
I think people should know, as early as possible, whether they have iron deficiency, even if they don’t have full-blown anemia. Once you know you are low in something, you can do something about it and start feeling better (or get back your nice, lush head of hair)!
These are the tests I recommend to check for both early signs and overt cases of anemia:
- Complete blood count (CBC) – A CBC can help gauge the severity of anemia. In chronic iron deficiency anemia, both the mean corpuscular volume (MCV) and the mean corpuscular hemoglobin concentration (MCHC) will be below normal. Reference range values for MCV and MCHC are 83-97 fL and 32-36 g/dL, respectively. You may also see that the platelet count is elevated (>450,000/µL), while the white blood cell (WBC) count is usually within reference ranges (4500-11,000/µL), but it may be elevated as well.
- Peripheral smear – This test involves examining your red and white blood cells and your platelets under a microscope, and can give insight into blood cell and platelet size, as well as any changes in the blood cells and platelets. Platelets are usually increased with anemia. This test can detect changes in the cells before MCV levels are decreased.
- Serum iron, total iron-binding capacity (TIBC), and serum ferritin – Low iron and ferritin levels with an elevated TIBC indicate iron deficiency. Low ferritin on its own is typically indicative of iron deficiency as well, but in some conditions, patients will have normal ferritin levels while still being iron deficient. These tests are useful in distinguishing iron deficiency anemia from other types of anemias.
Here is a chart that can be helpful to determine what stage of iron deficiency someone is in:
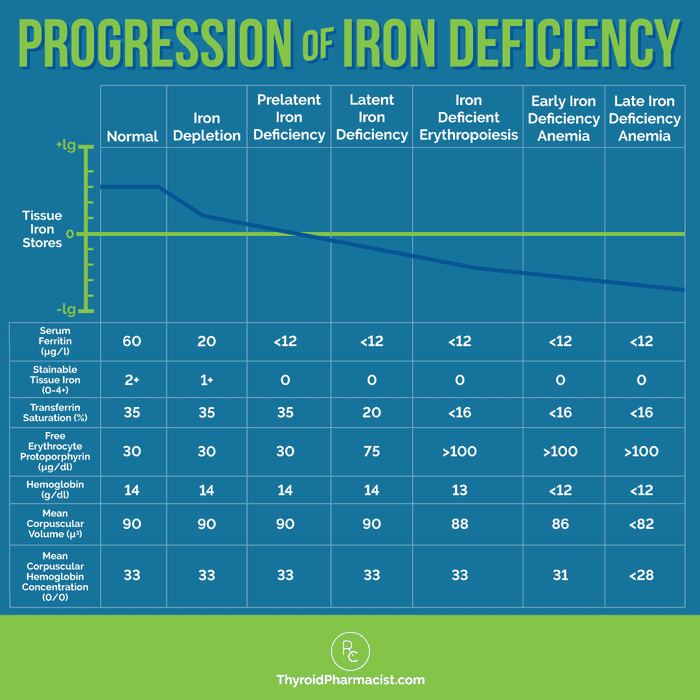
If you have a practitioner who is willing to work with you, that practitioner can order these tests for you. If you don’t have one, you can self-order the ferritin test, CBC, and iron/TIBC test through Ulta Labs.
I love Ulta Lab Tests for this purpose. You can self-order the labs and later submit them to your insurance for potential reimbursement (please check with your individual insurance to make sure they will cover the test).
Even if your insurance doesn’t cover it, the ferritin test is not expensive, and you can use your Health Savings Account in most cases.
Remember that ferritin represents your body’s iron reserve protein and is the best measure of how much iron your body is storing (i.e. how much iron is truly available for your body’s use).
Note that other tests for iron, or even tests for anemia, may indicate that you are within the normal reference range, but your ferritin levels may still be low (which is why I recommend the ferritin test).
We don’t want low levels of ferritin, as it is important in our body’s ability to utilize T3, which is necessary for our thyroid health. (When we have suboptimal T3 levels, we may experience hair loss, overall thinning, or patterns of baldness!)
Yet many people, especially women, are deficient in ferritin. Remember, ferritin is thought to be deficient in up to 50 percent of women of childbearing age, and women who are pregnant or postpartum are also at additional risk. Based on my experience, any woman with Hashimoto’s, or anyone having issues with hair loss, should take a ferritin test.
The normal ferritin lab range for women is between 12 and 150 ng/mL.
But again, we don’t want the normal range: we are always aiming for the optimal range. The optimal level for our thyroid is between 90-110 ng/mL.
I can tell you that personally, I was a teary, emotional, hair-losing mess when my ferritin was at 16 ng/mL! I was also cold all of the time, despite being on thyroid meds.
When it comes to hair loss, some experts feel that ferritin levels of at least 40 ng/mL are needed to stop hair loss, and that a level of at least 70 ng/mL is needed for hair regrowth.
Again, I don’t recommend supplementing with iron unless you’ve been tested and deemed as deficient. Too much iron/ferritin can be a problem. And as we’ve seen, even with supplementation, you need to address your root causes to prevent iron supply and demand challenges.
That’s why I recommend testing your ferritin levels before supplementing and retesting your levels within 1-3 months, to ensure that you are supplementing enough (but not too much!).
My Personal Anemia Story
Before I was diagnosed with Hashimoto’s, I personally had so much fatigue, constant bruising on my legs, bags under my eyes, and very pale skin first, and then the hair loss started shortly thereafter. I also have had a few other bouts with anemia that I had to resolve to feel fully human. 🙂

Here’s a photo of me the summer before my Hashimoto’s diagnosis. I was already a pharmacist, but I have to admit, my training in physical assessment was really lacking then. It’s now so obvious to me that my face was super pale and that I had bags under my eyes in that photo, despite it being the summer and spending time at the beach. Furthermore, my legs were covered in bruises. At the time, I remember thinking, “Why is it that my body gets so tan, but not my face?” for a while. I thought yellow just wasn’t my color because I looked so washed out. This was a trip to Poland, and I remember my auntie recommending arnica to me for my leg bruising. (By the way, if you have leg bruising, yes, it really works!)

In contrast, here’s a photo of me from last week, with healthy ferritin stores. And yes, I’m so much older (and so much wiser) – but also, my skin has a healthy color to it and I don’t have those bags under my eyes, despite having a 5 year old who wakes me up too early every now and then. 🙂 My legs don’t bruise easily anymore either. Interestingly, my face tans as well as my body when I spend time in the sun, and is the same skin tone that it was prior to puberty.
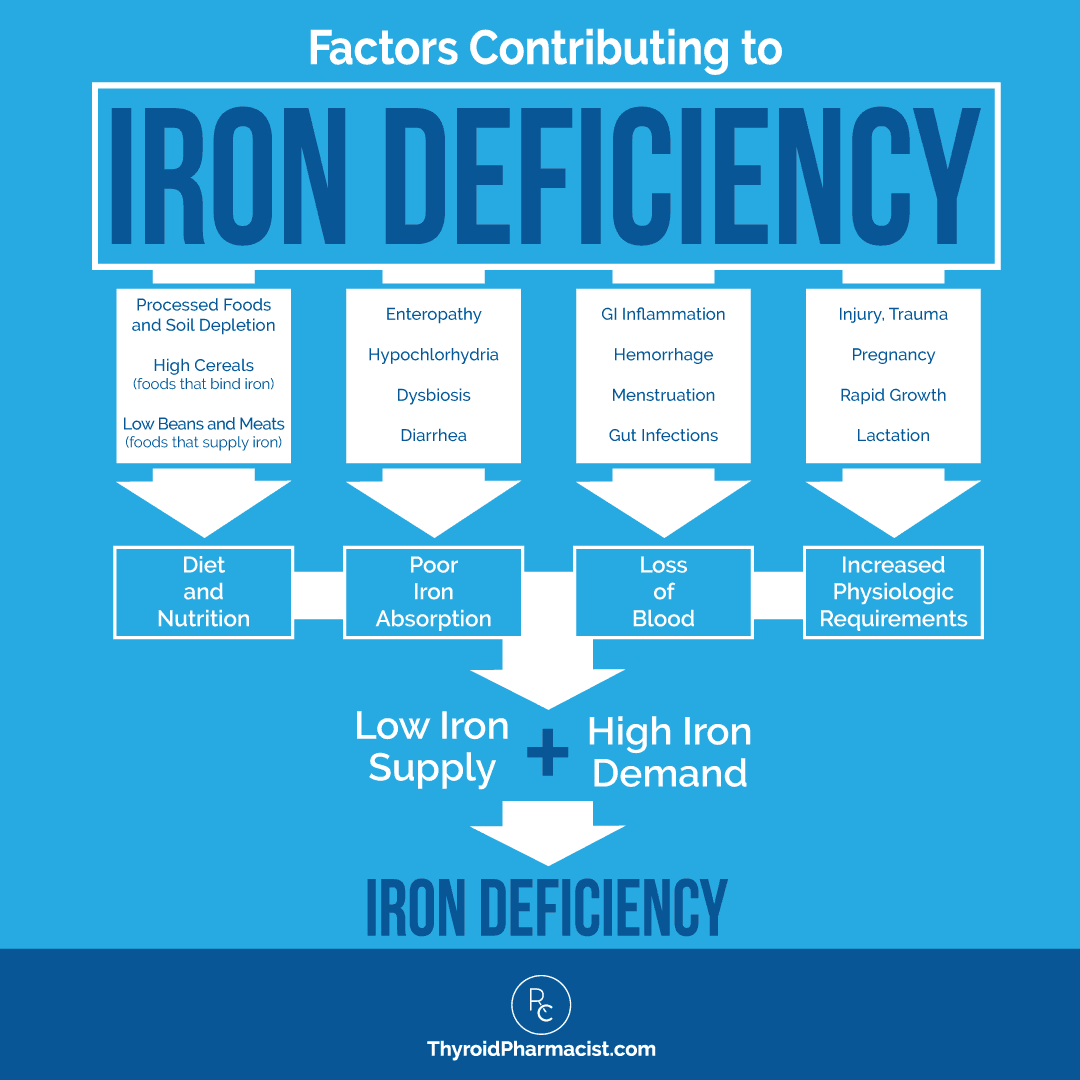
The Causes of Iron Deficiency In Hashimoto’s
So what causes low levels of ferritin, and why is iron deficiency anemia so common in Hashimoto’s?
I usually like to think of iron deficiency anemia in terms of iron supply and demand in the body. When you have higher than normal demands and/or a lower supply of iron, you are going to have deficiencies and symptoms.
When you look at iron in terms of supply and demand, it is easy to see why people with Hashimoto’s in particular often experience issues with iron deficiency; many of the issues that are related to iron status are also common triggers for Hashimoto’s.
The good news is that once you identify your root cause (supply/demand) issues and start to address them, you can typically improve your iron levels and feel better quickly.
Do You Have a Low Iron Supply?
On the iron “supply” side, we have the iron that we take in from our diet, along with the ability of our body to adequately extract and absorb that iron. Let’s review the ways our body gets iron, as well as the factors that impact our body’s ability to absorb the iron we consume.
Diet
Diet (as it often is!) is the first issue that could be affecting your iron supply.
Most of us do not get enough iron from our diets in the first place. Many of us are likely consuming several milligrams below the current recommended daily allowance (RDA) of 18 mg, so this makes our body’s ability to absorb the iron we do eat even more important.
Low dietary intake of iron-rich foods (such as meat and beans) can be one reason for not meeting the minimum intake. Vegetarians and vegans are more likely to experience low dietary intake of iron because they don’t eat meat, and plants provide a less absorbable form of iron, known as non-heme iron.
Heme iron is the form found in animal proteins, and also comprises 95 percent of the iron in our bodies. [15] It is better absorbed by the body than non-heme iron, which is typically found in plant sources like grains, nuts, seeds, legumes, and leafy greens. Eating heme iron from meat generally has the ability to boost iron levels much more than non-heme iron.
Other diets, such as high-fat diets, can also cause deficiencies, due to reduced iron absorption in the gut. [16]
Our public health officials wanted to solve common iron deficiency, and so iron-fortified cereals have been designed to help people achieve their minimum RDA of iron (but note that these types of cereals can often be highly processed and made from grains, such as gluten-containing ones, that are problematic for many people).
I’ll give you some ideas for what to eat later in this article, to ensure that you’re getting enough iron from foods, but beyond straight up supply, there are additional reasons you may be deficient in iron, such as malabsorption, excess blood loss, infections, and deficiencies in cofactor nutrients. [17]
Here are some factors that I have identified that may cause iron deficiency.
Celiac Disease
Celiac disease often co-occurs with Hashimoto’s, and can lead to iron deficiency. [18] Iron deficiency is frequently noted in patients with newly diagnosed celiac disease, and in some cases, it may be the only clinical feature that these patients display. [19]
The gut is compromised in celiac disease, and this can cause reduced nutrient absorption, including iron absorption. This is the main cause of iron deficiency in celiac disease; and less commonly, bleeding in the GI tract can contribute to iron deficiency. [20]
Removing gluten from the diet is one of the first interventions I suggest for those with Hashimoto’s. Even without celiac disease, many people find that removing gluten from their diet results in a dramatic improvement in thyroid symptoms.
In my survey of over 2000 people with Hashimoto’s, 88 percent of people who went gluten free felt better! And 86 percent of people who went gluten free reported an improvement in digestive symptoms. Notably, only 3.5 percent of survey respondents were diagnosed with celiac disease. This confirms what many of us in the world of functional medicine believe to be true: you do not have to have celiac disease to benefit from a gluten-free diet.
In many cases, people actually absorb nutrients more effectively from their food when they remove gluten from their diet! In fact, I’ve seen multiple cases of iron/ferritin deficiency resolve on a gluten-free diet.
Autoimmune Gastritis
Autoimmune gastritis is a chronic inflammatory disease that occurs when your immune system attacks your stomach lining. It frequently co-occurs in those with Hashimoto’s (along with celiac disease). [21]
The loss of parietal cells in the gut seen with autoimmune gastritis, causes reduced gastric acid secretion, which leads to impaired nutrient absorption, and iron deficiency is commonly found in those with autoimmune gastritis. [22]
Interestingly, autoimmune gastritis is often caused by H. pylori, a gut infection that is common in those with Hashimoto’s, and a trigger of autoimmune disease for many. [23] In this case, eradicating H. pylori should improve iron status (and potentially reduce or reverse symptoms associated with gastritis and Hashimoto’s).
H. pylori
Speaking of the devil, let’s talk about H. pylori – this is a notorious gut bug that can cause so many problems. When clients fill out their health timeline, they often identify symptoms that point to having one or more gut infections, including H. pylori. (In 2015, some 80 percent of my clients who took the gut tests that I recommended, tested positive for at least one gut infection!)
H. pylori is an important trigger for both Hashimoto’s and Graves’. I talked about this extensively in a previous article. It has also been linked to low stomach acid and gastritis, each associated with decreased iron absorption and lower iron levels. [24]
The bacteria that causes H. pylori has the ability to neutralize the acidity of its environment in the gut, which allows it to proliferate and survive. [25] This is what leads to low levels of stomach acid when someone has an H. pylori infection.
H. pylori also increases iron loss, as the infection needs to consume iron to sustain itself and grow – more on that when we talk about iron “demand”. [26]
H. pylori is also known to reduce vitamin C in the body, a vitamin which supports iron absorption, and H. pylori eradication has been shown to increase vitamin C levels as well as increase iron availability. [27]
Interestingly, premenopausal women seem to be at the greatest risk for iron deficiency anemia when they have an H. pylori infection, likely due to their having smaller iron stores to begin with (due to menstruation, a common demand that I will talk about in the next section).
Low Stomach Acid (Hypochlorhydria)
I know that many of you have low stomach acid, but might not recognize that you do. Raising my levels of stomach acid was actually a big breakthrough for me, that led me to getting my years of fatigue, acid reflux and Hashimoto’s into remission. We know from the research that low stomach acid is correlated with impacts on nutrient absorption, including iron, and with Hashimoto’s. [28] Did you know that acid reflux is actually a symptom of low stomach acid, and not high stomach acid? Not many people know this unfortunately, and are often prescribed acid-suppressing medications.
Additionally, using proton pump inhibitors (PPIs) and other acid suppressants can affect the absorption of iron. Iron needs acid present in order to be absorbed, so low stomach acid or taking antacids can reduce the iron supply that your body can absorb from your food (as well as any iron supplementation you might be taking)!
Low stomach acid is also a risk factor for developing bacterial conditions such as small intestinal bacterial overgrowth (SIBO), which has been found to be present in about 50 percent of those with Hashimoto’s. SIBO has been found to cause malabsorption issues with a variety of nutrients, including iron. [29]
Furthermore, low stomach acid has also been associated with other infections in the gut (such as H. pylori) and even parasites. [30]
When I started my own root cause investigation, I found that I had an H. pylori infection and low stomach acid, as well as anemia.
I was able to eradicate the infection and find successful protocols for addressing my low stomach acid (which I still use today). By treating H. pylori and supporting my stomach acid, I was able to improve my anemia, but my ferritin levels were still less than optimal for the longest time.
Heavy Metal Exposures
Toxic exposures are another known trigger for Hashimoto’s, in particular heavy metal toxicity such as toxic mercury levels. [31] Research has found that heavy metals can markedly alter the function and metabolism of many trace elements, including iron, copper, and selenium. [32] Heavy metals such as mercury can also increase the rate of red blood cell destruction, which depletes iron, as iron is stored in our blood.
Furthermore, heavy metal toxicity impacts other nutrients such as copper. [33] When our metabolism of copper is affected, our body can’t do as good a job in supporting iron absorption.
Gut Inflammation and Imbalances
A condition known as enteropathy (intestinal inflammation) has been associated with most gastrointestinal diseases, including intestinal infections, celiac disease, chronic diarrhea, irritable bowel syndrome, Crohn’s disease, and others. [34] Our gut microbiome plays a role in iron uptake and absorption, so compromised gut function can impair iron levels.[35]
There are various forms of enteropathy, one being gluten sensitive enteropathy. [36] We know that gluten is a problem for most people with Hashimoto’s and can result in gut inflammation for those who are sensitive to it.
Often, the primary symptom of enteropathy is iron deficiency anemia; sometimes with other digestive issues, sometimes not. Gluten sensitive enteropathy, for example, results in inflammation of the bowel surface, which reduces the bowel’s absorption of a variety of nutrients (including iron).
Finally, many of the common prescription medications (including the aforementioned acid blockers, as well as antibiotics, synthetic estrogens, and even birth control pills) we see as problematic for the thyroid, can also cause inflammation in the gut, further affecting our ability to extract nutrients from food, and result in nutrient deficiencies.
So, are you getting enough iron and its cofactors from your diet? And how well are you absorbing these nutrients?
Recommended Root Cause Testing When You Have Low Ferritin
Low ferritin can be connected to other conditions and infections, so if you’ve been addressing the causes listed above and are still having a hard time raising your ferritin levels, I suggest digging deeper with some root cause testing.
Given the high incidence of pathogens found in those with Hashimoto’s, I recommend working with your functional practitioner to also get a comprehensive stool analysis, like the GI-MAP (GI Microbial Assay Plus) – Diagnostic Solutions Kit. This test checks for parasites, bacteria, viruses, and fungi in the gut. It also measures bacterial balance of normal flora and digestive markers, like elastase and secretory IgA, which provide further insight on overall digestive and intestinal health. Note that this test needs to be ordered through your practitioner’s account or via Direct Labs self-ordering; if you’re self-ordering, it likely won’t be covered by insurance, but you’ll need to check.
If your doctor won’t order the tests, I’d suggest finding a new practitioner who is functionally inclined. A good functional medicine practitioner is more likely to review potential root causes in your health history, as well as listen to what you have to say about what makes you feel better and what makes you feel worse.
They will search for infections (such as H. pylori) or other relevant health issues (such as your use of acid blockers) to dig for possible root causes as well.
Does Your Body Have High Iron Demands?
Once iron is absorbed, it is stored for future use as ferritin in the liver, skeletal muscle, spleen, duodenum, marrow, and elsewhere in the body. There, it is available as the body needs it.
You may think of your body’s iron demand in terms of blood loss, such as with menstruation, injury, hemorrhaging, transfusions, etc. While this type of obvious blood loss is part of your body’s iron demand, there is a lot more to it than that. But let’s start with menstruation, as that is a high iron demand period in women’s lives (pun intended).
Menstruation
Menstruating women lose blood every month. If your periods are too heavy or too frequent, you may lose more blood than you’re able to take in. There is a potential loss of 1 mg of iron per cycle for women with normal periods, and as much as 5-6 mg per cycle for women with heavy periods. [37] This is one reason the prevalence of iron deficiency is higher in premenopausal women – we lose iron every month, and some of us more than others, especially if we have hormonal imbalances!
Hormonal imbalances, like estrogen dominance, can also cause very heavy periods where the body loses too much iron, as can low levels of progesterone, PCOS, and perimenopause.
Heavy periods may also be a sign that your thyroid hormone dosage may need to be adjusted, as being hypothyroid can also cause a heavy flow.
Furthermore, I have observed that some women tend to have heavy periods with lots of blood clots when they have an MTHFR gene mutation, but are not properly supported with adequate amounts of methylfolate (1,000 – 5,000 mcg per day is the range to consider). See my article on MTHFR to learn more.
Oftentimes, iron deficiency and heavy periods can be a vicious cycle. Your periods can actually become too heavy because you are iron deficient, and with each cycle, you lose more blood, and become even more iron deficient.
Ladies, I have a confession – I was almost 40 years old when I realized that my periods were too heavy, and that the giant blood clots in my menses were not normal.
In case you’ve been in the dark like I have been, here’s what a normal period looks like:
A regular menstrual cycle occurs once every 21-35 days, and lasts for 3-7 days on average. Some women have blood clots in their period blood, and clots smaller than the size of a quarter are usually not cause for concern. [38] The average woman loses about 60 milliliters (2.7 ounces) of blood during her period, and can be expected to soak one to seven normal-sized pads or tampons during her entire period. [39] (Of course, you will often change a pad/tampon before it’s fully soaked.)
Heavy periods are considered to entail over 80 milliliters of blood loss. If you’re going through more than nine to twelve regular-sized pads or tampons during your cycle, you may have heavy periods. [40] This was a shock to me, as I was usually soaking through numerous tampons with each cycle, until I worked on my periods.
Depending on the cause of your heavy menses, you may need iron support, proper thyroid hormone levels, methylation support (in my experience, this is helpful for giant blood clots/heavy bleeding), myo-inositol (for normalizing period frequency and flow), progesterone, Vitex, or seed cycling support (I love Beeya for an easy way to incorporate seed cycling into my daily life), to turn your heavy periods into normal periods.
I love my friend Jolene’s book Is this Normal?, which talks about some things that many of us don’t think to ask about, that have to do with our hormones and menstrual cycles. I also love the book Period Repair Manual by Lara Briden and Fix Your Period by Nicole Jardin.
Some Less Common Sources and Solutions to Consider
Gastrointestinal Blood Loss
If you are a long-distance runner or endurance athlete, you may be at a higher risk for iron deficiency, especially if you’re a woman!
This problem is significantly underreported, and most runners and athletes aren’t aware of the risk.
A 1989 study measured serum ferritin levels of high school cross country runners, and found that 34 percent of female runners and 8 percent of male runners had ferritin levels below 12 ng/mL, which we know is really low… so imagine all the other runners who may have had suboptimal levels! [41]
Female runners may be especially susceptible to iron deficiency. A 2020 study found that 28 percent of female marathon runners had iron deficiency, compared to only 1.6 percent of male marathon runners.
This is because endurance athletes and long distance runners may experience blood loss in the gastrointestinal tract. A 1996 study out of Germany found that periods of intensive training or racing were associated with a three- to six-fold increase in GI blood loss. [42]
In a smaller 2022 study, 13 non-professional female athletes were evaluated for iron deficiency. Six of the 13 women (about 46 percent) were found to have iron levels below 30 ng/mL. [43]
Blood loss from GI bleeding, in addition to blood loss during menstruation, makes female runners and endurance athletes especially susceptible to iron deficiency.
Another clue you may be iron deficient is if you feel like your athletic performance has decreased – maybe you’re feeling fatigued earlier in your training, or struggling on runs or exercises that previously felt do-able to you.
If you’re an endurance athlete, I would recommend testing your ferritin levels.
Sweating
If you are someone who sweats a lot, either naturally, because of hyperhidrosis (excessive sweating not related to heat or exercise), because you’re very active, or because you use the sauna often – you may be at higher risk of iron deficiency! This is because we lose iron through our sweat.
The average and daily loss of iron through sweat is about 1 mg per day for men, and 1.4 mg per day for women, with about 500 mL per day of sweat production. In hyperhidrosis, the rate of sweat production can get up to 12 L per day, resulting in a loss of iron ranging from 4.6 mg to 8.3 mg per day! [44]
So if you’re sweating a lot regularly, you may be at a higher risk of iron deficiency. In addition to the reported GI blood loss that can play a role in iron deficiency seen in endurance athletes, such as marathon and distance runners, I think that excess sweating may also play a role in why iron deficiency is so common in these types of athletes.
Anemia Due to Viral Infections
Viral infections have the potential to cause anemia due to their effect on bone marrow. Viruses that have been associated with anemia include Epstein-Barr Virus, hepatitis A and C, human herpes virus 6 (HHV-6), cytomegalovirus, parvovirus B19, and HIV. [45] Epstein-Barr in particular is associated with Hashimoto’s, and may be some people’s trigger.
In fact, a 2015 Polish study found the Epstein-Barr virus in the thyroid cells of 80 percent of people with Hashimoto’s, while controls did not have EBV present in their thyroid gland. [46]
In this case, we can look to the essential amino acid lysine for support. Lysine serves two important functions in this scenario. Not only has lysine been used to control viral infections, but it has also been shown to increase iron absorption.
Lysine has long been used to control herpes simplex virus (HSV), and studies have shown that improving lysine levels led to a reduction of HSV attacks, fewer healing days, and milder symptoms. [47] This is because lysine can actually suppress viral replication. It’s also been shown to block other viruses like the one we shall not speak of, and the influenza A virus.
When it comes to iron, lysine appears to support the uptake of iron. Researchers have found that supplementation with daily iron in women experiencing hair loss, resulted in only some women experiencing a modest increase in serum ferritin concentration. However, when they added 1.5 g per day of lysine to their existing iron supplementation, there was a significant increase in mean serum ferritin concentrations, and the women experienced a 39 percent decrease in hair shedding after six months of treatment. [48]
For anyone who has both a viral infection and anemia, lysine may be an excellent two-for-one tool.
Physiological Changes in the Body
Any type of physiological change such as pregnancy, lactation, or even a period of rapid growth, can affect the body’s need for additional iron.
Iron deficiency anemia can occur during pregnancy due to a higher demand for iron. [49] During the initial six months of a woman’s pregnancy, the fluid portion of her blood can increase faster than the number of red blood cells can. This dilutes the blood and can lead to anemia during pregnancy.
This happened to me. No amount of eating beef or taking supplements would help! During my pregnancy, I had what my hubby and I called a burger deficiency, where I would cry and be sad if I didn’t eat exactly two grass-fed burgers a day. 🙂
Despite all of the burgers, like many women, I found I had to have iron infusions (which work very quickly, do not cause constipation, and can restore ferritin levels in as little as a few weeks), several times throughout my pregnancy, to address a wide range of symptoms (including dizziness, insomnia, fatigue, anxiety, restless legs, mood swings, and crying)!
Similarly, iron may be depleted while a woman is lactating, as milk production requires iron (among many other nutrients). When a woman is coming out of the pregnancy stage already depleted in her iron stores, followed by the birthing process (which often results in blood loss and further iron depletion), she can enter the demanding phase of breastfeeding with already low iron stores. Lactation can then further deplete those iron stores, which may result in a lowered milk supply.
Dr. Oscar Serrallach, author of The Postnatal Depletion Cure, has mentioned that low iron is one of the most common postpartum depletions he sees in his practice, that can lead to women having health issues post-baby.
Parasitic Worms
Like infections, worms and other parasites can affect both supply and demand for our body’s iron. On the supply side, research has shown that worms such as hookworms can cause poor absorption of many essential nutrients, including iron. [50] Worms can also increase demand due to causing blood loss. Worm infections related to hookworms (Necator americanus, Ancylostoma duodenale, Strongyloides stecoralis, Giardia lamblia), whipworms (Trichuris trichiura), and roundworms (Ascoris lumbricoides) have all been tied to iron deficiency, either through causing poor absorption, causing the destruction of red blood cells, or causing gastrointestinal bleeding. [51]
Hookworms can cause blood loss by attaching to the intestinal mucosa, tearing into capillaries and ingesting the blood.
I know the thought of worms and other parasites may literally make your skin crawl, but many people have infections, viruses, and parasites (I did, too). You’ll see my recommendations in the testing section for evaluating whether or not these types of infections may be one of your root causes.
Gastrointestinal Pathogens
Many of the aforementioned critters and other pathogens need iron to sustain themselves and grow. In fact, there is research that shows that too much iron (iron overload) can increase our susceptibility to infections, and allow existing infections to thrive. [52]
Our bodies have developed mechanisms to withhold iron from these types of “bad” microorganisms: iron-binding proteins (such as ferritin) work to decrease iron reutilization, reduce iron absorption, and reduce the production of red blood cells… therefore decreasing iron availability for unwanted pathogens. [53]
This protective mechanism is part of a natural inflammatory response that occurs in our gut. If the body perceives there is a pathogenic attack of some kind, it is going to do all it can to block access to iron. This inflammatory response can cause people many significant gastrointestinal symptoms and conditions, including iron deficiency anemia. [54]
This notion of using iron as a way to combat pathogens has led to a whole investigation of how iron deficiency could be used in the treatment of certain diseases. Given today’s challenge of increasing antibiotic resistance, we are starting to see how manipulation of iron stores in the body could be one way to “starve” off a given infection in the future. Note that it also means that just popping iron pills is not a viable strategy, as it could prove to be life-sustaining for pathogens. This is another reason why it is important to know and monitor your ferritin levels.
Anemia of Chronic Kidney Disease
Anemia is a common complication of chronic kidney disease (CKD). [55] Interestingly, hypothyroidism is highly prevalent in CKD, and a growing body of evidence suggests that hypothyroidism may be a risk factor for developing CKD, and worse health outcomes for those with CKD. [56]
Anemia in CKD can be due to several factors. When the kidneys are damaged, they produce less erythropoietin (EPO), which is a hormone that tells your bone marrow to produce red blood cells. Additionally, the red blood cells in people with anemia and CKD tend to have a shorter lifespan in the bloodstream, and these cells die faster than they can be replaced. People with CKD and anemia may also be low in levels of critical nutrients that are needed to make healthy red blood cells like iron, vitamin B12, and folate. [57]
In addition to supplementing with iron and any other vitamins someone may be deficient in, treatment for anemia in CKD typically consists of an erythropoiesis-stimulating agent (ESA), which sends a message to your bone marrow to produce more red blood cells. Patients on hemodialysis may be prescribed IV or subcutaneous ESAs during dialysis treatments. In more advanced cases of anemia, blood transfusions may be used.
Just this year, the FDA approved Jesduvroq tablets (daprodustat) as the first oral treatment for anemia caused by chronic kidney disease, for adults who have been receiving dialysis for at least four months. [58] While shown to be effective for raising hemoglobin levels, it also comes with potential side effects like high blood pressure, thrombotic vascular events, abdominal pain, dizziness, and allergic reactions.
These treatment options don’t work well for everyone, and in the case of blood transfusions, may come with complications like the body producing antibodies against the donor blood cells.
Arginine is an alternative treatment option that has been shown to raise hemoglobin levels with few to no side effects. One study took a group of eight elderly patients with anemia associated with kidney disease. [59] The researchers gave the patients 1.3 grams per day of L-arginine and found that all eight patients had increased hemoglobin levels, and six of the patients even showed improved kidney function. More impressively, there were no reported side effects.
Citrulline is a precursor to arginine, and so may be beneficial for those with anemia as well. Citrulline may be more easily absorbed than arginine, and has been shown to be effective in increasing L-arginine levels. [60]
The Root Cause Approach to Improving Iron Deficiency
Low iron/ferritin levels, and the issue of hair loss and lack of energy, can often be addressed via dietary changes and supplementation, but you may also have to look underneath the surface to discover other things that might be going on. Is there something causing a high iron demand (heavy periods? GI Issues?)?
When I work with a client, I will look at their overall symptoms, medical history, and test results; this will usually tell me if iron deficiency is an issue. My root cause approach will also typically highlight other issues we’ll need to work on, such as addressing low stomach acid, toxicity, infections, or parasites.
I love to start with food as medicine for cutting down the branches, while we dig for the root cause. The great thing about starting with food (and simple digestive support) is that you can often feel better fairly quickly. You can also start on dietary changes while you look for a good functional practitioner if you don’t already have one.
Food as Medicine (Mmmmm… liver!)
Dietary iron can be found in two forms: heme iron and non-heme iron. Heme iron is derived almost exclusively from meat (beef, pork, veal, chicken, lamb, turkey, and others), fish, and seafood.
Non-heme iron is most frequently found in vegetables (wheat, barley, corn, legumes, corn, cooked spinach, dried fruits, nuts, seeds, and others), but also in iron-fortified foods such as bread, pasta, rice, cereals, and dairy products. (You’ll notice that many of these are foods that people with Hashimoto’s consider to be food sensitivities!)
The absorption of iron from these two forms can vary greatly. Heme iron (from meat) is the better-absorbed version, with an absorption rate of 20 to 30 percent. This is why vegetarians and vegans, in particular, need to ensure they are getting adequate iron from their more limited diet.
I recommend working on restoring iron/ferritin levels by eating sources of heme iron, like organ meats, which are particularly high in iron. I usually recommend eating cooked liver or beef a few times per week to begin with, unless your dietary belief goes against that.
Liver is a rich source of iron, but I realize that many people don’t like it.
A “cheat” way is to take desiccated liver in supplement form such as the PaleoValley Extract or Dessicated Liver made by Allergy Research Group. Please note: liver is also high in other nutrients such as copper (which is needed for iron absorption, but people with copper toxicity may need to avoid liver supplements).
You may also wish to try this recipe of mine. 🙂
Liver is one of the richest sources of iron and may be helpful in restoring iron and ferritin levels, but it is not most people’s favorite. Even if you haven’t liked liver in the past, I hope you’ll try this Liver Pâté recipe I make at home – it’s full of other bold flavors that help balance out the richness of the liver.
Liver Pâté Recipe
Prep time: 10 minutes
Cook time: 5 to 7 minutes
Serves: 8
Note: It is helpful to eat a serving with a cup of hot lemon water to improve digestion.
Ingredients:
1 pound beef, chicken, or pork liver, cut into chunks
1 onion, chopped
1 tablespoon duck fat or coconut oil
1 tablespoon coconut fat from coconut milk
1 garlic clove, crushed
¼ cup fresh basil, chopped
4 teaspoons apple cider vinegar
Juice from ½ lemon
1 tablespoon ground cinnamon
Sea salt or pink Himalayan sea salt, to taste
Black pepper, to taste (if tolerated)
Pickles, for garnish
Directions:
1. Fry the liver and onion in a pan with the duck fat or coconut oil for about 5 to 7 minutes, until cooked through.
2. Place the liver and onion along with the coconut fat, garlic, basil, vinegar, lemon juice, and cinnamon in a food processor. Blend until smooth. Season with salt and pepper to taste.
3. Form the mixture into a ball and chill for 1 hour.
4. To serve, place in a bowl or slice in thick slices, and garnish with pickles.
Adding vitamin-C rich foods to your iron-containing meals (such as broccoli to a steak dinner), can help with extracting iron from the foods.
Additionally, adding beetroot to your routine can also help. Beets are a rich source of vitamin C and betaine, both of which support iron absorption. A study of anemic teen girls found that two weeks of drinking beetroot juice helped with increasing iron levels. [61]
Chlorophyll, a green pigment contained in plants and a bioavailable source of copper, has also been found to raise iron levels. You can add chlorophyll to green smoothies for a daily boost of iron! Please keep in mind that chlorophyll can be an issue for those with copper toxicity (covered in detail in my Hashimoto’s Self Management Program).
There are also a few things you should avoid consuming alongside iron-containing foods to ensure proper absorption.
For example, tannins in tea and coffee should be spaced out by an hour from iron-containing meals. [62] Phytates found in nuts, legumes, and grains, as well as egg whites, may also affect iron absorption. Furthermore, iron needs an acid present for optimal absorption. Avoid antacids and calcium supplements around meal-times. Lastly, remember to space thyroid medications at least four hours away from iron-rich meals!
Iron-Boosting Routines
There are simple routines we can incorporate into our everyday lives that can boost our iron levels as well. One of my favorite ways to do that is through what we cook with!
Cast iron skillets are made of iron, and cooking in them can actually raise iron levels. Depending on the food, the iron content can be increased by as many as six to eight milligrams.
I also love the Lucky Iron Fish, which is a cute little reusable cooking tool made from food-grade electrolytic iron powder. You simply drop the lucky fish into any boiling liquid with some acidity, and it releases iron – super easy and fun! It can be a great way to add some iron into veggie rich soups and stews.
Supplementing with Iron Directly
Many people with anemia or low ferritin may be advised to take iron supplements by their physicians or pharmacists.
And yes, iron supplementation can help! It’s something that I have often recommended to clients, and in my survey of more than 2232 people with Hashimoto’s, 81 percent saw improvement in energy from ferritin/iron supplementation, so it is definitely something to consider once you know your levels are low.
People often ask me what my favorite iron supplements are, and if all iron pills are created equally.
There are various iron salts, or compounds, and each of them have different levels of elemental iron in them. [63] I remember dispensing ferrous sulfate most often in the community pharmacy, but ferrous gluconate and ferrous fumarate are some other examples of the types that are available over the counter.
I personally have heard a lot of feedback from clients that most over-the-counter iron pills cause or worsen constipation. I have found that iron bisglycinate is the most gentle on the stomach and is least likely to cause constipation. If you’re looking for a brand recommendation, Thorne’s Iron Bisglycinate is the one I have most commonly used for people with anemia.
As iron supplements are the most common cause of toxicity, please keep out of reach of children and pets, and please work with your doctor or pharmacist for proper lab and weight-based dosing. Yes, the dosing may be different depending on the person!
According to the Society of the Advancement of Patient Blood Management, “therapeutic doses of iron should increase hemoglobin levels by 0.7-1.0 g/dL per week”, and “adequate iron replacement has typically occurred when the serum ferritin level reaches 100 µg/L.” [64]
Iron supplements are best taken on an empty stomach (food impairs absorption), and iron supplements need to be taken four hours apart from thyroid meds!
It’s important to note that many people don’t properly absorb iron from supplements (due to things like calcium supplements, acid-suppressing medications, drinking tea, having H. pylori, and of course, the various gut issues that present with Hashimoto’s), and some people are unable to tolerate the therapeutic doses of iron due to the side effects. [65]
Some clients find that taking an iron supplement every other day works better for them, while others have upped their dosage of magnesium citrate to resolve iron-induced constipation. (BTW, my constipation article is coming soon!)
Some additional alternatives that my clients have found to be better absorbed/less constipating include:
Please be sure to work with your doctor or pharmacist to ensure you are getting proper testing and dosing of your iron supplements.
Addressing Anemia and Ferritin With Gut Issues
When I was trying to raise my ferritin levels, I was doing all of the things, but I personally didn’t see a big change in my levels. I was baffled, until I learned that for people with gut issues or gut infections, taking iron orally is not just less effective, but can further worsen dysbiosis, because pathogenic bacteria use the iron to build biofilms! Biofilms are a collective of microorganisms that grow on biological surfaces and often house gut pathogens, which can, in turn, lead to infections.
When we have gut issues or infections, we may not be able to absorb iron from our food or supplements very well. Now this can be a tricky situation, because we want optimal iron and ferritin levels, but we don’t want to make any potential gut issues worse!
Back in 2016, I had the honor of being a Keynote Speaker at a Functional Medicine Conference in Poland, and had the pleasure of speaking with numerous experts on their approaches to Hashimoto’s. I was fascinated to learn about aloe and lactoferrin at that conference. Both were touted by the functional nutritionists as incredibly helpful for clients with Hashimoto’s. (See my article on aloe that I released a few years back to learn more.)
Lactoferrin is an iron absorption-regulating protein found in mammalian milk. Through binding the iron, it makes it unusable for the pathogenic organisms, while allowing us to use it. It also acts as a biofilm buster, helping to resolve gut infections. Lactoferrin was compared to ferrous sulfate and was found to be more effective at raising serum iron, hemoglobin, and ferritin levels, while being better tolerated, leading to less GI distress. [66]
Lactoferrin not only “starves” pathogenic microbes of iron, but also promotes the growth of beneficial bacteria like Lactobacillus and Bifidobacteria. [67]
A study in pregnant women found that lactoferrin, at a dose of 250 mg per day, was more effective at raising hemoglobin levels AND produced much less constipation as well (note to self if I choose to have another baby, as that pregnancy constipation is NOT something I remember fondly, haha). [68]
Furthermore, lactoferrin may produce less inflammation compared to elemental iron, and might actually be helpful for anemia due to chronic inflammation. [69]
Based on its efficacy, safety, and tolerability profile, a 2017 review article that looked at all of the studies done on lactoferrin vs. elemental iron (including fetal outcome studies), actually recommended that lactoferrin should be the treatment of choice for pregnant women with iron deficiency! [70]
Additionally, researchers in Egypt determined that lactoferrin is a more safe and effective way of treating iron deficiency anemia in children. [71]
It’s best taken on an empty stomach or right before a meal. Doses of 100-400 mg daily, for up to 12 weeks, are generally well-tolerated. I have used and recommended Jarrow Formulas, and their supplement offers a 250 mg dose per capsule.
One important thing to be aware of when it comes to lactoferrin is that it is derived from whey, so if you’re sensitive to dairy, you’ll want to consider how it may affect you. The main constituents are α-lactalbumin (α-LA), β-lactoglobulin (β-LG) and lactoferrin (LF), which account for approximately 70–80 percent of total whey protein. The β-lactoglobulin is the most allergenic, but there is emerging technology that allows companies to isolate the lactoferrin and remove the β-lactoglobulin. [72] AOR Lactoferrin-250 is one such product.
Lactoferrin can also be found in colostrum. Colostrum supplements are gaining popularity recently for their benefit for the gut and the immune system. [73] They also contain lactoferrin, and can be helpful in boosting ferritin levels. Colostrum is the first form of breastmilk produced after giving birth, and colostrum supplements are typically derived from bovine sources. Keep this in mind if you are sensitive to dairy. Pure Encapsulations has a colostrum supplement, as does Designs for Health.
Interestingly, while lactoferrin levels are present in freshly expressed milk, the pasteurization process removes most of the lactoferrin from milk products. [74]
Iron Infusions
Another option, especially for those who have tried EVERYTHING, is to work with a practitioner to obtain intravenous iron. Intravenous iron can restore your ferritin levels with a few IVs that can be done in two weeks time, won’t give you constipation, and won’t require you to take a daily pill.
Iron infusions can also help bypass the gut, and studies have shown that iron infusions will not impact gut dysbiosis. [75]
This is a great option for people who are not absorbing iron orally. While any doctors can prescribe or order iron IVs, most clinics are not set up to offer them.
Look for integrative, functional, and naturopathic clinics that offer this service, or a medical specialist that is known as a hematologist.
Additional Supplements That Support Your Body’s Iron Supply
I often suggest increasing one’s vitamin C intake, as that increases one’s absorption of iron. You can take a vitamin C supplement, or just increase your intake of vitamin C-rich foods when eating an iron-rich meal. (Broccoli is a great one that I love! You can eat a serving of organic steak with a hearty serving of broccoli.)
If you feel like you are eating enough red meat or liver, ask yourself if your underlying root cause could be low stomach acid. Low stomach acid can contribute to low iron levels and anemia by affecting nutrient absorption, as stomach acid is needed to extract iron and vitamin B12 from our protein-containing foods.
Symptoms of low stomach acid can include: acid reflux, low ferritin or low B12 levels, fatigue despite thyroid medications, as well as constipation or diarrhea. Many people with Hashimoto’s have this issue, which can often be easily corrected by taking enzyme support such as Betaine with Pepsin. This supplement changed my life!
Additionally, Herbal Bitters and thiamine can help the body produce its own stomach acid when taken over time.
Along with digestive enzymes, probiotics containing Lactobacillus plantarum have helped my clients increase their iron absorption by as much as 50 percent. [76] (I recommend Probiotic 50B.) For more information on how probiotics can help with Hashimoto’s, you can read my article on the best probiotics for Hashimoto’s.
Precautions
Iron can build up in your body, and taking too much can be deadly. Iron supplements are a leading cause of overdose for children, as well as adults, which is why there is a need for both testing and retesting! For this reason, I recommend retesting your levels within one to three months of starting supplementation.
Some Additional Things I’m Exploring
Lately, as I thought I was done with the rabbit hole, I came across iron cofactors and iron recycling. Iron cofactors have been popularized by Dr. Ben Lynch, and iron recycling by health researcher Morley Robbins. Both concepts are similar, but use a different approach.
Iron Cofactors
A recent deep dive of mine has focused on what my colleague Dr. Ben Lynch calls “cofactors” – supportive nutrients that assist with the body’s ability to absorb and utilize iron.
Dr. Lynch has identified a number of cofactors that are necessary to ensure efficient absorption and utilization of iron, including vitamin C, vitamin A, riboflavin (B2), biotin, and copper.
Vitamin C is known for its ability to enhance absorption of iron through a process in the intestinal tract that creates a form of iron that’s easier for the body to absorb. [77]
Low vitamin A tends to be associated with low iron status, and the combination of vitamin A and iron is thought to reduce iron deficiencies more quickly than iron alone. [78] This may be due to vitamin A’s ability to transport and distribute iron in the body, and it may also help iron mobilize from storage in the body.
Riboflavin (vitamin B2) is an essential B vitamin, and deficiencies may impair iron absorption, increase intestinal loss of iron, and impair iron utilization. [79] In a 2011 study, 119 women that had a riboflavin deficiency were given either 2 g or 4 g of riboflavin, or a placebo for eight weeks. [80] In the groups who supplemented, there was a significant increase in hemoglobin status. There were no changes to their iron intake through food or supplements during that time, suggesting the importance of riboflavin in iron absorption.
I like Riboflavin by Integrative Therapeutics, and often for people with anemia-related fatigue, headaches, or migraines, I will recommend 400-800 mg per day.
Biotin is another essential B vitamin that’s necessary for heme production, and deficiency appears to lower heme levels in cells. [81]
Lastly, copper supports the movement of iron out of its ferritin stores. [82]
For those of us with Hashimoto’s, we may be deficient in multiple important nutrients given our propensity toward leaky gut, infections, and other digestion/GI issues. This is one reason why some people may not get enough iron, even with a supplement.
Some people may need some cofactor support as they tackle the root causes of their low iron supplies (or higher demands).
I personally think vitamin C, riboflavin, and biotin are excellent and safe for most people, but recommend using caution with copper supplementation, and only using it under the supervision of a practitioner who is well versed in copper toxicity, as some people can develop copper toxicity! Dr. Lynch has create a product specially formulated to include iron’s cofactors, Optimal Iron.
Iron Recycling
Further down the rabbit hole, I came across iron recycling. This is a theory popularized by health researcher Morley Robbins and hair tissue mineral analysis (HTMA) proponents that focus on using magnesium, vitamin A in the retinol form, and bioavailable copper, to ”recycle” the iron.
According to iron recycling proponents, our body produces 24 mg of iron every 24 hours, and we need 25 mg of iron per day to produce red blood cells. Our body’s production of iron depends on this iron recycling system, where iron stored in our tissues is liberated into our serum.
This is important because we want iron to be in active transport in our bodies, and when we have a build up of iron in our tissues, this drives inflammation and many associated symptoms.
In order for iron to move from these tissues into circulation, HTMA proponents report that we need key nutrients, including magnesium, vitamin A (retinol), and bioavailable copper. By strategically supplementing with these nutrients, HTMA proponents claim that we can ensure that iron is able to move from the tissue to the serum and be “recycled” for use in the body.
Registered dietician Christa Beigler, who is an HTMA expert, recommends Epsom salt baths for magnesium, cod liver oil for the retinol form of vitamin A, and chlorophyll for bioavailable copper.
I don’t have much experience with HTMA yet, although I do find it fascinating, so I will be taking a course on HTMA and trying this out. I thought about finishing the HTMA course and testing this theory out with a few hundred clients before I published this article, but that would probably add a few years to the editing time of this article (which is already at almost 10 years, haha), so I decided that I will just update this article when I learn more.
My Personal Ferritin Journey
In my personal experience, I have struggled with low ferritin levels for a super long time.
I have also had low levels of hemoglobin/hematocrit (a more advanced type of anemia).
I was able to normalize the hemoglobin/hematocrit with iron/ferritin supplements, eating red meat, treating gut infections including H. pylori, taking betaine with pepsin and digestive enzymes, eating liver, taking liver pills, cod liver oil, and chlorophyll from 2012 to 2015. That said, my ferritin levels did not budge, despite the same methods mentioned above working for others!
I continued to struggle with low ferritin levels and even ended up with copper toxicity, so I stopped taking liver pills, cod liver oil, and chlorophyll, as they’re all possible sources of copper. Copper toxicity is another article I have been working on for almost eight years, but it is covered in detail in my Hashimoto’s Self-Management Program.
Rather than trying to increase my ferritin levels, I used thyroid medications containing T3 as a workaround to bypass the system. Using T3 medications directly meant that I didn’t need iron for the T4 to T3 conversion to happen, and I avoided most of the symptoms associated with low ferritin.
It wasn’t until I got pregnant that I felt an urgency to address my low ferritin levels, as iron deficiency in pregnancy is associated with premature birth, low birth weight, and increased maternal illness. [83]
Because my ferritin was so low, my physician recommended iron infusions, and I found many of the “common symptoms of pregnancy” like insomnia, tearfulness, and restless legs vanished within the first infusion!
My ferritin levels were stable postpartum, and I had lactational amenorrhea for a very long time. When my periods did come back, they were lighter and regular, until I had a dreaded health flare in 2020, when my son was a little over two years old.
This health flare occurred in my late thirties and caused me to have super heavy periods twice per month, which led to me draining my ferritin stores once more. 🙁
The health flare happened right when the pandemic started, and I think the cause was likely stress, taking care of a young child 24/7, coming off all of my supplements, toxic exposure, and transitioning from eating a high protein/low carb diet to skipping breakfast, chugging my thyroid meds with coffee, and subsisting on toddler leftovers for three months. 😉
During my health flare, I drained my iron stores and needed to get iron infusions. This restoration of iron helped to make my periods less heavy. I also needed an adjustment in thyroid meds to make sure my TSH was between 0.5 – 2 μIU/mL, but my frequent cycles persisted.
I did a lot of detective work to get back into a healthy cycle and tried various things, including topical progesterone for perimenopause, which did help, but I would start bleeding if I ever missed a dose!
I also gave up coffee, as caffeine/coffee is tied to excess menstrual bleeding and shorter cycles – and this seemed to help, but sometimes I would get my period early right after drinking coffee, and sometimes my period would still come super early, even when I didn’t have any coffee. [84]
Something just didn’t sit well with me with this approach, so I also tried repeating my Liver Reset because I had toxic exposure, and took P5P and Vitex to help with regulating my hormones – all of these were super helpful.
My personal protocol to keep my periods regular is eating for blood sugar balance, eating a high-protein diet, taking thyroid meds on an empty stomach, reducing caffeine intake (no more than three cups of black tea per day), taking two capsules per day of MTHFR Pathways, taking 2000 mg of myo-inositol per day (which is actually closer to a PCOS dose of myo-inositol than the Hashimoto’s dose of 600 mg), and trying Beeya seed cycling.
Interestingly, I was really stable healthwise during and for two years after my pregnancy, and I think part of the reason is that I was properly supplementing. But then I came off all of my supplements a few months before I had my health flare.
Before that, I had been taking 1000 mcg of methylfolate and 600 mg of myo-inositol (the typical dose for Hashimoto’s) per day with excellent results, but after coming off and going back on, I had to increase my dosage of both to get back into balance. I watched my menstrual clots vanish when I increased my methylfolate to about 3400 mcg per day, and my periods became regular and lighter with using myo-inositol at a 2000 mg dose.
High Ferritin – Iron Overload/Toxicity
As a side rabbit hole, while I had been struggling with low ferritin that was discovered in 2011 and resolved in 2017 (it also flared up in 2020, then again in 2022), ironically, my husband had iron overload that was diagnosed in 2018, and it resolved in 2021 due to some extreme lifestyle changes. I am happy to report that both of our iron levels are now in the optimal range.
Ferritin above 200 ng/mL in women (and 300 ng/mL in men) can be an indication of iron toxicity/overload.
Symptoms may include:
- Extreme fatigue
- Brain fog
- Weakness
- Heart flutters or irregular heartbeat
- Joint and stomach pain
- Irritability
- Depression
Iron toxicity is more common in men, postmenopausal women, people with genes for hemochromatosis Types I, II, III, IV, and people living at elevations above 5,000 feet.
Iron toxicity can also be the result of excess inflammation, in which case, resolving the source of inflammation can help reduce iron levels.
The standard of care for high iron/ferritin levels is therapeutic phlebotomies every few months. Interestingly, elevation can affect our iron levels as well. If you live at a higher elevation, moving to sea level can be curative within three to six months of moving for elevation-induced cases.
In 2018, my husband and I hunkered down in Boulder, CO before the birth of our son, and he started having some very odd symptoms, including insomnia, ankle pain, and overwhelm. At first we thought that it was just worries about being a new parent, but I realized something was off when we found out his liver enzymes were elevated. We dug deeper and realized he had high ferritin and iron overload.
He started therapeutic phlebotomies and felt much better almost immediately after the first phlebotomy, and for a few weeks after each phlebotomy… but the symptoms would restart again, and tests revealed that his ferritin levels would quickly rise. 🙁
I was like, “Really, why can’t I just have some of your ferritin?!”
Oddly enough, he didn’t have the genes for hemochromatosis 1, 2, or 3 (done by standard tests), but he did have the gene for type 4, which we found through some advanced genetic testing. This is not the usual hemochromatosis gene, but rather, this genetic variation prevented him from efficiently removing iron from his body. Essentially, he was making more iron but not removing it quickly enough.
I am thankful that my mom (retired MD and RN, and co-author of Hashimoto’s: The Root Cause), helped me track down some studies of how high altitude hypoxia could lead to iron overload. [85]
At high altitudes, the body responds to the low oxygen (hypoxia) by producing more of a hormone called erythropoietin, which stimulates the production of red blood cells to help with transporting the oxygen. Under normal circumstances, the excess iron is carried out by ferroportin, the iron exporter molecule. However, when someone has type 4 hemochromatosis (which is more common than the other types), it affects ferroportin function, and excess iron can accumulate in tissues. [86]
Essentially, my husband was accumulating the iron due to the low oxygen and not moving it out because of a ferroportin gene mutation.
We went on vacation to Los Angeles, CA in 2019, when our son was a little over a year old, and something strange happened during the two weeks we were on vacation – my husband slept just fine, and most of his iron overload symptoms vanished. We thought it was a fluke of just being more relaxed on vacation, but my gut told me we needed to be at sea level. We traveled back and forth to LA numerous times in 2019 and 2020, with my hubby feeling so much better each time.
I kept campaigning for a move, as it became obvious to me that the altitude wasn’t working well for my husband (and also, the altitude seemed to affect our son’s sleep as well, so that meant none of us were sleeping), and he finally agreed to a trial of being in LA for at least three months in December of 2020. His iron overload and all of the associated symptoms completely vanished within three months.
I’m happy to report that his ferritin levels have been within the optimal range every time, and he has not needed therapeutic phlebotomies since.
While we loved our home and our loved ones in Colorado – we really thought it was going to be our forever home – we don’t plan on living at high altitudes again. But if we did, or if someone had ongoing iron overload issues, I would recommend they look into the following options for iron overload, in addition to therapeutic phlebotomies:
- Pure Encapsulations IP6 (Inositol Hexaphosphate) may lower ferritin and reduce the need for therapeutic phlebotomies. [87] Please note that you may need to supplement with calcium and zinc, as IP6 can lower them too. Doses may vary.
- Milk thistle can help protect the liver from damage caused by excess iron. I recommend Pure Encapsulations Milk Thistle at a dose of 250 mg, one to four times per day.
- Lactoferrin – So get this! Not only does lactoferrin help raise ferritin levels when they’re too low, but lactoferrin can also help lower high levels of ferritin! [88]
- Lowering your intake of dietary iron, as well as eating phytates and oxalates with iron-rich foods, may be helpful for some, but it is not usually curative and can also prevent the absorption of other nutrients such as zinc and calcium.
As always, please discuss with your practitioner before taking new supplements. They know what underlying health issues you have, as well as what medications or other supplements you are taking, and can advise against any contraindications.
Anemia of Inflammation
Last but not least, I’ve had clients that have reached out to me with odd lab numbers. An individual may have some numbers that suggest iron deficiency, and others that suggest iron overload, leaving them confused – are they iron deficient or not? This type of anemia tends to be more common in people with chronic illness, infections, and autoimmunity, and researchers have used the term “anemia of inflammation” to describe this phenomenon where some people might have normal/high ferritin, but still have anemia.
According to the NIH, “In anemia of inflammation, you may have a normal or sometimes increased amount of iron stored in your body tissues, but a low level of iron in your blood. Inflammation may prevent your body from using stored iron to make enough healthy red blood cells, leading to anemia.” [89]
Again, interestingly, lactoferrin is something that may be able to correct this!
Next Steps
I know this may seem like a lot to take in, but please don’t feel overwhelmed! The good news is that many of the same root causes for iron deficiency (and related symptoms like hair loss) are also root causes of Hashimoto’s… so know that you are addressing multiple conditions by doing the same set of interventions.
Begin by looking at your symptoms and your health timeline. When discussing anemia symptoms with a functional medicine practitioner, it is important for them to know if you may have low stomach acid, SIBO, or an H. pylori infection, as all of these factors can affect the absorption of iron and other key nutrients.
For more information on how to optimize your ferritin levels with Hashimoto’s, you can download a free copy of my Supplements to Subdue Thyroid Symptoms eBook. Be sure to also check out the nutrient depletions guide in my cookbook, Hashimoto’s Food Pharmacology: Nutrition Protocols and Healing Recipes to Take Charge of Your Thyroid Health!
I hope this helps, and I wish you all the best on your health journey!
I love interacting with my readers on social media, and I encourage you to join my Facebook, Instagram, TikTok, and Pinterest community pages to stay on top of thyroid health updates and meet others who are following similar health journeys. For recipes, a FREE Thyroid Diet start guide, and notifications about upcoming events, be sure to sign up for my email list!
P.S. Please note, I don’t make or sell lactoferrin, but I wanted to get the word out to you that this is something that is potentially going to be very helpful, as I was excited about the research and results people have been getting. For whatever reason, one of the brands I used to recommend for this has just raised their prices on PureRXO, the dispensary I usually link to. You can get access to various brands of lactoferrin via my Fullscript dispensary, with my discount. Unfortunately, Fullscript doesn’t allow people to view any products until they make an account. You can sign up here to view my recommended products.
References
[1] Dutt S, Gupta P, Singh U. Association of Serum Ferritin, Folate, Vitamin B12 with Thyroid Hormone Levels in Patients with Thyroid Disorders. J Assoc Physicians India. 2023;71(1):1.
[2] Dutt S, Gupta P, Singh U. Association of Serum Ferritin, Folate, Vitamin B12 with Thyroid Hormone Levels in Patients with Thyroid Disorders. J Assoc Physicians India. 2023;71(1):1.
[3] Eftekhari MH, Keshavarz SA, Jalali M, Elguero E, Eshraghian MR, Simondon KB. The relationship between iron status and thyroid hormone concentration in iron-deficient adolescent Iranian girls. Asia Pac J Clin Nutr. 2006;15(1):50-55.
[4] Soliman AT, De Sanctis V, Yassin M, Wagdy M, Soliman N. Chronic anemia and thyroid function. Acta Biomed. 2017;88(1):119-127. Published 2017 Apr 28. doi:10.23750/abm.v88i1.6048
[5] Galton VA. Some effects of altitude on thyroid function. Endocrinology. 1972;91(6):1393-1403. doi:10.1210/endo-91-6-1393; Surks MI. Effect of thyrotropin on thyroidal iodine metabolism during hypoxia. Am J Physiol. 1969;216(2):436-439. doi:10.1152/ajplegacy.1969.216.2.436
[6] Rayman MP. Multiple nutritional factors and thyroid disease, with particular reference to autoimmune thyroid disease. Proc Nutr Soc. 2019;78(1):34-44. doi:10.1017/S0029665118001192
[7] Soliman AT, De Sanctis V, Yassin M, Wagdy M, Soliman N. Chronic anemia and thyroid function. Acta Biomed. 2017;88(1):119-127. Published 2017 Apr 28. doi:10.23750/abm.v88i1.6048
[8] Cinemre H, Bilir C, Gokosmanoglu F, Bahcebasi T. Hematologic effects of levothyroxine in iron-deficient subclinical hypothyroid patients: a randomized, double-blind, controlled study. J Clin Endocrinol Metab. 2009;94(1):151-156. doi:10.1210/jc.2008-1440; Ravanbod M, Asadipooya K, Kalantarhormozi M, Nabipour I, Omrani GR. Treatment of iron-deficiency anemia in patients with subclinical hypothyroidism. Am J Med. 2013;126(5):420-424. doi:10.1016/j.amjmed.2012.12.009
[9] Rayman MP. Multiple nutritional factors and thyroid disease, with particular reference to autoimmune thyroid disease. Proc Nutr Soc. 2019 Feb;78(1):34-44.
[10] Abu-Ouf NM, Jan MM. The impact of maternal iron deficiency and iron deficiency anemia on child’s health. Saudi Med J. 2015;36(2):146-149. doi:10.15537/smj.2015.2.10289
[11] Maguire JL, Salehi L, Birken CS, et al. Association between total duration of breastfeeding and iron deficiency. Pediatrics. 2013;131(5):e1530-e1537. doi:10.1542/peds.2012-2465
[12] R V, Dhiman P, Kollipaka R, P S, V H. Association of Hypothyroidism With Low Serum Ferritin Levels and Iron-Deficiency Anemia During the First Trimester of Pregnancy. Cureus. 2022;14(8):e28307. Published 2022 Aug 23. doi:10.7759/cureus.28307
[13] Freeman AM, Rai M, Morando DW. Anemia Screening. In: StatPearls. Treasure Island (FL): StatPearls Publishing; July 25, 2022.
[14] Waldbieser J. The Scary Mental Health Risks of going meatless. Women’s Health. December 2, 2015. Accessed July 9, 2023. https://www.womenshealthmag.com/food/a19941229/side-effects-of -vegetarianism/.
[15] Hooda J, Shah A, Zhang L. Heme, an essential nutrient from dietary proteins, critically impacts diverse physiological and pathological processes. Nutrients. 2014;6(3):1080-1102. Published 2014 Mar 13. doi:10.3390/nu6031080
[16] Sonnweber T, Ress C, Nairz M, et al. High-fat diet causes iron deficiency via hepcidin-independent reduction of duodenal iron absorption. J NutrBiochem. 2012 Dec;23(12):1600-8.
[17] Soliman AT, De Sanctis V, Yassin M, Wagdy M, Soliman N. Chronic anemia and thyroid function. Acta Biomed. 2017;88(1):119-127. Published 2017 Apr 28. doi:10.23750/abm.v88i1.6048
[18] Starchl C, Scherkl M, Amrein K. Celiac Disease and the Thyroid: Highlighting the Roles of Vitamin D and Iron. Nutrients. 2021;13(6):1755. Published 2021 May 21. doi:10.3390/nu13061755; Rayman MP. Multiple nutritional factors and thyroid disease, with particular reference to autoimmune thyroid disease. Proc Nutr Soc. 2019;78(1):34-44. doi:10.1017/S0029665118001192
[19] Freeman HJ. Iron deficiency anemia in celiac disease. World J Gastroenterol. 2015;21(31):9233-9238. doi:10.3748/wjg.v21.i31.9233
[20] Ibid
[21] Rayman MP. Multiple nutritional factors and thyroid disease, with particular reference to autoimmune thyroid disease. Proc Nutr Soc. 2019;78(1):34-44. doi:10.1017/S0029665118001192
[22] Kulnigg-Dabsch S. Autoimmune gastritis. Autoimmungastritis. Wien Med Wochenschr. 2016;166(13-14):424-430. doi:10.1007/s10354-016-0515-5
[23] Veijola LI, Oksanen AM, Sipponen PI, Rautelin HI. Association of autoimmune type atrophic corpus gastritis with Helicobacter pylori infection. World J Gastroenterol. 2010;16(1):83-88. doi:10.3748/wjg.v16.i1.83
[24] Smolka AJ, Schubert ML. Helicobacter pylori-Induced Changes in Gastric Acid Secretion and Upper Gastrointestinal Disease. Curr Top Microbiol Immunol. 2017;400:227-252. doi:10.1007/978-3-319 -50520-6_10; Annibale B, Capurso G, Delle Fave G. The stomach and iron deficiency anaemia: a forgotten link. Dig Liver Dis. 2003;35(4):288-295. doi:10.1016/s1590-8658(03)00067-7; Hershko C, Ronson A, Souroujon M, et al. Variable hematologic presentation of autoimmune gastritis: age-related progression from iron deficiency to cobalamin depletion. Blood. 2006 Feb 15;107(4):1673-9.
[25] Berger A. Scientists discover how helicobacter survives gastric acid. BMJ. 2000;320(7230):268.
[26] Monzón H, Forné M, Esteve M, et al. Helicobacter pylori infection as a cause of iron deficiency anaemia of unknown origin. World J Gastroenterol. 2013;19(26):4166-4171. doi:10.3748/wjg.v19.i26.4166
[27] Mei H, Tu H. Vitamin C and Helicobacter pylori Infection: current knowledge and future prospects. Front Physiol. 2018;9:1103.; Haile K, Yemane T, Tesfaye G, Wolde D, Timerga A, Haile A. Anemia and its association with Helicobacter pylori infection among adult dyspeptic patients attending Wachemo University Nigist Eleni Mohammad Memorial Referral Hospital, Southwest Ethiopia: A cross-sectional study. PLoS One. 2021;16(1):e0245168. Published 2021 Jan 14. doi:10.1371/journal.pone.0245168; Vitale G, Barbaro F, Ianiro G, et al. Nutritional aspects of Helicobacter pylori infection. Minerva Gastroenterol Dietol. 2011 Dec;57(4):369-77.
[28] Guilliams TG, Drake LE. Meal-Time Supplementation with Betaine HCl for Functional Hypochlorhydria: What is the Evidence?. Integr Med (Encinitas). 2020;19(1):32-36.; Daher R, Yazbeck T, Jaoude JB, Abboud B. Consequences of dysthyroidism on the digestive tract and viscera. World Journal of Gastroenterology. 2009;15(23):2834-2838. doi:10.3748/wjg.15.2834; Jacobs A, Miles PM. Role of gastric secretion in iron absorption. Gut. 1968;10(3), 226.
[29] Bures J, Cyrany J, Kohoutova D, et al. Small intestinal bacterial overgrowth syndrome. World Journal of Gastroenterology. 2010;16(24):2978-2990.; Sachdev AH, Pimentel M. Gastrointestinal bacterial overgrowth: pathogenesis and clinical significance. Therapeutic Advances in Chronic Disease. 2013;4(5):223-231.
[30] Tilahun M, Gedefie A, Belayhun C, Sahle Z, Abera A. Helicobacter pylori Pathogenicity Islands and Giardia lamblia Cysteine Proteases in Role of Coinfection and Pathogenesis. Infect Drug Resist. 2022;15:21-34. Published 2022 Jan 6. doi:10.2147/IDR.S346705
[31] Rezaei M, Javadmoosavi SY, Mansouri B, Azadi NA, Mehrpour O, Nakhaee S. Thyroid dysfunction: how concentration of toxic and essential elements contribute to risk of hypothyroidism, hyperthyroidism, and thyroid cancer. Environ Sci Pollut Res Int. 2019;26(35):35787-35796. doi:10.1007/s11356-019- 06632-7
[32] Tchounwou PB, Yedjou CG, Patlolla AK, Sutton DJ. Heavy metal toxicity and the environment. Exp Suppl. 2012;101:133-164. doi:10.1007/978-3-7643-8340-4_6
[33] Collins JF, Prohaska JR, Knutson MD. Metabolic crossroads of iron and copper. Nutr Rev. 2010;68(3):133–147.
[34] Gibbons T, Fuchs GJ. Chronic enteropathy: clinical aspects. Nestle Nutr Workshop Ser Pediatr Program. 2007;59:89-104. doi:10.1159/000098529; Levitt DG, Levitt MD. Protein losing enteropathy: comprehensive review of the mechanistic association with clinical and subclinical disease states. Clin Exp Gastroenterol. 2017;10:147–168.
[35] Qi X, Yun C, Pang Y, Qiao J. The impact of the gut microbiota on the reproductive and metabolic endocrine system. Gut Microbes. 2021;13(1):1-21. doi:10.1080/19490976.2021.1894070; Yilmaz B, Li H. Gut Microbiota and Iron: The crucial actors in health and disease. Pharmaceuticals (Basel). 2018;11(4):98.
[36] Zamani F, Mohamadnejad M, Shakeri R, et al. Gluten sensitive enteropathy in patients with iron deficiency anemia of unknown origin. World J Gastroenterol. 2008;14(48):7381–7385.
[37] Napolitano M, Dolce A, Celenza G, et al. Iron-dependent erythropoiesis in women with excessive menstrual blood losses and women with normal menses. Ann Hematol. 2014;93(4):557-563. doi:10.1007/s00277-013-1901-3
[38] Brighten J. Phases of the Menstrual Cycle. Dr. Jolene Brighten. July 9, 2021. Accessed July 10, 2023. https://drbrighten.com/phases-of-the-menstrual-cycle/.
[39] Heavy periods: Overview . National Library of Medicine. 2017. Accessed July 10, 2023. https://www.ncbi.nlm.nih.gov/books/NBK279294/.; Prior JC. Very heavy menstrual flow. The Centre for Menstrual Cycle and Ovulation Research. October 4, 2017. Accessed July 12, 2023. https://www.cemcor.ubc.ca/resources/very-heavy-menstrual-flow.
[40] Prior JC. Very heavy menstrual flow. The Centre for Menstrual Cycle and Ovulation Research. October 4, 2017. Accessed July 12, 2023. https://www.cemcor.ubc.ca/resources/very-heavy-menstrual-flow.
[41] Nickerson HJ, Holubets MC, Weiler BR, Haas RG, Schwartz S, Ellefson ME. Causes of iron deficiency in adolescent athletes. J Pediatr. 1989;114(4 Pt 1):657-663. doi:10.1016/s0022-3476(89)80717-6
[42] Nachtigall D, Nielsen P, Fischer R, Engelhardt R, Gabbe EE. Iron deficiency in distance runners. A reinvestigation using Fe-labelling and non-invasive liver iron quantification. Int J Sports Med. 1996;17(7):473-479. doi:10.1055/s-2007-972881
[43] Sims ST, Mackay K, Leabeater A, Clarke A, Schofield K, Driller M. High Prevalence of Iron Deficiency Exhibited in Internationally Competitive, Non-Professional Female Endurance Athletes-A Case Study. Int J Environ Res Public Health. 2022;19(24):16606. Published 2022 Dec 10. doi:10.3390/ijerph192416606
[44] Labib M, Obeid D. Hyperhidrosis and iron deficiency. Ann Clin Biochem. 1995;32 ( Pt 5):509-510. doi:10.1177/000456329503200514
[45] Morinet F, Leruez-Ville M, Pillet S, Fichelson S. Concise review: Anemia caused by viruses. Stem Cells. 2011;29(11):1656-1660. doi:10.1002/stem.725
[46] Janegova A, Janega P, Rychly B, Kuracinova K, Babal P. Rola infekcji wirusem Epstein-Barr’a w rozwoju autoimmunologicznych chorób tarczycy. Endokrynologia Polska. 2015;66(2):132-136. doi:10.5603/ep.2015.0020.
[47] Melano I, Kuo LL, Lo YC, Sung PW, Tien N, Su WC. Effects of Basic Amino Acids and Their Derivatives on SARS-CoV-2 and Influenza-A Virus Infection. Viruses. 2021;13(7):1301. Published 2021 Jul 4. doi:10.3390/v13071301
[48] Rushton DH. Nutritional factors and hair loss. Clin Exp Dermatol. 2002;27(5):396-404. doi:10.1046/j.1365-2230.2002.01076.x
[49] Abu-Ouf NM, Jan MM. The impact of maternal iron deficiency and iron deficiency anemia on child’s health. Saudi Med J. 2015;36(2):146-149. doi:10.15537/smj.2015.2.10289; Li S, Gao X, Wei Y, et al. The relationship between iron deficiency and thyroid function in chinese women during early pregnancy. J Nutr Sci Vitaminol (Tokyo). 2016;62(6):397-401.
[50] Seidelman J, Zuo R, Udayakumar K, et al. Caught on capsule: Iron-deficiency anemia due to hookworm infection. Am J Med. 2016 Feb;129(2):167-9.
[51] Hall A, Hewitt G, Tuffrey V, de Silva N. A review and meta-analysis of the impact of intestinal worms on child growth and nutrition. Matern Child Nutr. 2008;4 Suppl 1(Suppl 1):118-236. doi:10.1111/j.1740-8709.2007.00127.x
[52] Cassat JE, Skaar EP. Iron in infection and immunity. Cell Host Microbe. 2013;13(5):509-519. doi:10.1016/j.chom.2013.04.010
[53] Cronin SJF, Woolf CJ, Weiss G, Penninger JM. The Role of Iron Regulation in Immunometabolism and Immune-Related Disease. Front Mol Biosci. 2019;6:116. Published 2019 Nov 22. doi:10.3389/fmolb.2019.00116
[54] Wessling-Resnick M. Iron homeostasis and the inflammatory response. Annu Rev Nutr. 2010;30:105–122.
[55] Atkinson MA, Warady BA. Anemia in chronic kidney disease. Pediatr Nephrol. 2018;33(2):227-238. doi:10.1007/s00467-017-3663-y
[56] Rhee CM. The interaction between thyroid and kidney disease: an overview of the evidence. Curr Opin Endocrinol Diabetes Obes. 2016;23(5):407-415. doi:10.1097/MED.0000000000000275
[57] Anemia in chronic kidney disease. National Institute of Diabetes and Digestive and Kidney Diseases. September 2020. Accessed June 23, 2023. https://www.niddk.nih.gov/health-information/kidney- disease/anemia.
[58] FDA approves first oral treatment for anemia caused by chronic kidney disease for adults on dialysis. FDA. February 1, 2023. Accessed June 29, 2023. https://www.fda.gov/news-events/press- announcements/fda-approves-first-oral-treatment-anemia-caused-chronic-kidney-disease-adults-dialysis.
[59] Tarumoto T, Imagawa S, Kobayashi M, Hirayama A, Ozawa K, Nagasawa T. L-arginine administration reverses anemia associated with renal disease. Int J Hematol. 2007;86(2):126-129. doi:10.1532/IJH97.06212
[60] Allerton TD, Proctor DN, Stephens JM, Dugas TR, Spielmann G, Irving BA. l-Citrulline Supplementation: Impact on Cardiometabolic Health. Nutrients. 2018;10(7):921. Published 2018 Jul 19. doi:10.3390/nu10070921
[61] Purba RB, Paruntu OL, Ranti IN, et al. Beetroot juice and red spinach juice to increase hemoglobin levels in anemic adolescent girls. Open Access Macedonian Journal of Medical Sciences. September 17, 2021. Accessed July 16, 2023. https://oamjms.eu/index.php/mjms/article/view/6871#
[62] Morck TA, Lynch SR, Cook JD. (1983). Inhibition of food iron absorption by coffee. The American journal of clinical nutrition. 37(3), 416-420.
[63] Physician’s Guide to Oral Iron Supplements. Society for the Advancement of Patient Blood Management. December 29, 2020. Accessed July 18, 2023. https://sabm.org/physician-guide-to-oral-iron/.
[64]Ibid
[65] Alleyne M, Horne MK, Miller JL. Individualized treatment for iron-deficiency anemia in adults. Am J Med. 2008;121(11):943-948. doi:10.1016/j.amjmed.2008.07.012
[66] Zhao X, Zhang X, Xu T, Luo J, Luo Y, An P. Comparative Effects between Oral Lactoferrin and Ferrous Sulfate Supplementation on Iron-Deficiency Anemia: A Comprehensive Review and Meta-Analysis of Clinical Trials. Nutrients. 2022;14(3):543. Published 2022 Jan 27. doi:10.3390/nu14030543
[67] Vega-Bautista A, de la Garza M, Carrero JC, Campos-Rodríguez R, Godínez-Victoria M, Drago-Serrano ME. The Impact of Lactoferrin on the Growth of Intestinal Inhabitant Bacteria. Int J Mol Sci. 2019;20(19):4707. Published 2019 Sep 23. doi:10.3390/ijms20194707
[68] Rezk M, Dawood R, Abo-Elnasr M, Al Halaby A, Marawan H. Lactoferrin versus ferrous sulphate for the treatment of iron deficiency anemia during pregnancy: a randomized clinical trial. J Matern Fetal Neonatal Med. 2016;29(9):1387-1390. doi:10.3109/14767058.2015.1049149
[69] Paesano R, Berlutti F, Pietropaoli M, et al. Lactoferrin efficacy versus ferrous sulfate in curing iron deficiency and iron deficiency anemia in pregnant women. Biometals. 2010;23(3):411-417. doi:10.1007/s10534-010-9335-z; Lepanto MS, Rosa L, Cutone A, Conte MP, Paesano R, Valenti P. Efficacy of Lactoferrin Oral Administration in the Treatment of Anemia and Anemia of Inflammation in Pregnant and Non-pregnant Women: An Interventional Study. Front Immunol. 2018;9:2123. Published 2018 Sep 21. doi:10.3389/fimmu.2018.02123
[70] Abu Hashim H, Foda O, Ghayaty E. Lactoferrin or ferrous salts for iron deficiency anemia in pregnancy: A meta-analysis of randomized trials. Eur J Obstet Gynecol Reprod Biol. 2017;219:45-52. doi:10.1016/j.ejogrb.2017.10.003
[71] El-Khawaga A, Abdelmaksoud H. Effect of lactoferrin supplementation on iron deficiency anemia in primary school children. International Journal of Medical Arts. July 2019. Accessed July 18, 2023. https://ijma.journals.ekb.eg/article_34024.html.
[72] Isolation of lactoferrin from milk. Google Patents. August 24, 1995. Accessed June 27, 2023. https://patents.google.com/patent/US5861491A/en.; 1. Wu M, Xu Y. Isolation and purification of lactoferrin and immunoglobulin G from bovine colostrum with serial cation-anion exchange chromatography. SpringerLink. May 17, 2009. Accessed June 27, 2023. https://link.springer.com/article/10.1007/ s12257-008-0159-4.
[73] Chandwe K, Kelly P. Colostrum Therapy for Human Gastrointestinal Health and Disease. Nutrients. 2021;13(6):1956. Published 2021 Jun 7. doi:10.3390/nu13061956
[74] Paulaviciene IJ, Liubsys A, Eidukaite A, Molyte A, Tamuliene L, Usonis V. The Effect of Prolonged Freezing and Holder Pasteurization on the Macronutrient and Bioactive Protein Compositions of Human Milk. Breastfeed Med. 2020;15(9):583-588. doi:10.1089/bfm.2020.0219
[75] Puga AM, Samaniego-Vaesken ML, Montero-Bravo A, Ruperto M, Partearroyo T, Varela-Moreiras G. Iron Supplementation at the Crossroads of Nutrition and Gut Microbiota: The State of the Art. Nutrients. 2022;14(9):1926. Published 2022 May 4. doi:10.3390/nu14091926
[76] Hoppe M, Önning G, Berggren A, Hulthén L. Probiotic strain Lactobacillus plantarum 299v increases iron absorption from an iron-supplemented fruit drink: a double-isotope cross-over single-blind study in women of reproductive age. Br J Nutr. 2015;114(8):1195-202.
[77] Lynch SR, Cook JD. Interaction of vitamin C and iron. Ann N Y Acad Sci. 1980;355:32-44. doi:10.1111/j.1749-6632.1980.tb21325.x; Lane DJ, Richardson DR. The active role of vitamin C in mammalian iron metabolism: much more than just enhanced iron absorption! Free Radic Biol Med. 2014 Oct;75:69-83.
[78] da Cunha MSB, Campos Hankins NA, Arruda SF. Effect of vitamin A supplementation on iron status in humans: A systematic review and meta-analysis. Crit Rev Food Sci Nutr. 2019;59(11):1767-1781. doi:10.1080/10408398.2018.1427552
[79] Hilary J Powers, Riboflavin (vitamin B-2) and health, The American Journal of Clinical Nutrition, Volume 77, Issue 6, June 2003, Pages 1352–1360.
[80] Powers HJ, Hill MH, Mushtaq S, Dainty JR, Majsak-Newman G, Williams EA. Correcting a marginal riboflavin deficiency improves hematologic status in young women in the United Kingdom (RIBOFEM). Am J Clin Nutr. 2011;93(6):1274-1284. doi:10.3945/ajcn.110.008409
[81] Atamna H, Newberry J, Erlitzki R, Schultz CS, Ames BN. Biotin deficiency inhibits heme synthesis and impairs mitochondria in human lung fibroblasts. J Nutr. 2007;137(1):25-30. doi:10.1093/jn/137.1.25
[82] Chan WY, Rennert OM. The role of copper in iron metabolism. Ann Clin Lab Sci. 1980;10(4):338-344.
[83] Georgieff MK. Iron deficiency in pregnancy. Am J Obstet Gynecol. 2020;223(4):516-524. doi:10.1016/j.ajog.2020.03.006
[84] Fenster L, Quale C, Waller K, et al. Caffeine consumption and menstrual function. Am J Epidemiol. 1999;149(6):550-557. doi:10.1093/oxfordjournals.aje.a009851; 1. Mahmoud AZB, Makhdoom AN, Mufti L, Alreheli RS, Farghal RG, Aljaouni SE. Association between menstrual disturbances and habitual use of caffeine. ScienceDirect. December 2014. Accessed July 12, 2023. https://www.sciencedirect.com/science/article/pii/S1658361214000651.
[85] Zhang Y, Fang J, Dong Y, et al. High-Altitude Hypoxia Exposure Induces Iron Overload and Ferroptosis in Adipose Tissue. Antioxidants (Basel). 2022;11(12):2367. Published 2022 Nov 29. doi:10.3390/antiox11122367; Muckenthaler MU, Mairbäurl H, Gassmann M. Iron metabolism in high-altitude residents. J Appl Physiol (1985). 2020;129(4):920-925. doi:10.1152/japplphysiol.00019.2020; Bock E. Gassmann lectures on high-altitude physiology. National Institutes of Health. October 5, 2016. Accessed July 16, 2023. https://nihrecord.nih.gov/2018/10/05/gassmann-lectures-high-altitude-physiology.
[86] De Domenico I, Ward DM, Nemeth E, et al. The molecular basis of ferroportin-linked hemochromatosis. Proc Natl Acad Sci U S A. 2005;102(25):8955-8960. doi:10.1073/pnas.0503804102
[87] Bhowmik A, Ojha D, Goswami D, et al. Inositol hexa phosphoric acid (phytic acid), a nutraceuticals, attenuates iron-induced oxidative stress and alleviates liver injury in iron overloaded mice. Biomed Pharmacother. 2017;87:443-450. doi:10.1016/j.biopha.2016.12.125
[88] Graudal N, Milman N, Hegnhøj J, Galløe A. Has Lactoferrin a Role in Hereditary Hemochromatosis?. Hematology. 1997;2(5):421-422. doi:10.1080/10245332.1997.11746364; Schaible UE, Collins HL, Priem F, Kaufmann SH. Correction of the iron overload defect in beta-2-microglobulin knockout mice by lactoferrin abolishes their increased susceptibility to tuberculosis. J Exp Med. 2002;196(11):1507-1513. doi:10.1084/jem.20020897; Habib HM, Ibrahim S, Zaim A, Ibrahim WH. The role of iron in the pathogenesis of COVID-19 and possible treatment with lactoferrin and other iron chelators. Biomed Pharmacother. 2021;136:111228. doi:10.1016/j.biopha.2021.111228
[89] Anemia of Inflammation or Chronic Disease . National Institute of Diabetes and Digestive and Kidney Diseases. September 2018. Accessed July 18, 2023. https://www.niddk.nih.gov/health-information/blood- diseases/anemia-inflammation-chronic-disease.



